|
Although elements have some qualities in common, such as boiling points, melting points, and electronegativities, no two elements have exactly the same combination of properties. A sodium atom isn’t the same as a carbon atom. We still know that this is generally true now. He also pointed out that the atoms of one element are not the same as those of all other elements. Part 2: All atoms of a given element are identical in mass and properties.Įvery atom of an element, such as gold, is the same as every other atom of that element, according to Dalton. While this paradigm is useful in some situations, we now know that atoms are not solid spheres. Dalton’s atom could be seen as a piece in a molecular modelling kit, with different elements represented by spheres of various sizes and colours. He proposed that all matter is made up of atoms, which he described as “solid, weighty, hard, impenetrable, moving particle(s).”ĭalton did not have the proper apparatus to see or experiment on individual atoms, therefore he had no way of knowing if they had any interior structure. Dalton’s atomic theoryĪccording to Dalton, the law of conservation of mass and the law of definite proportions may be explained using the concept of atoms. By mixing sodium metal with chlorine gas, we may make more table salt with the same composition. Table salt, for example, has the molecular formula NaCl and has the same quantities of sodium and chlorine regardless of how much salt you have or where it comes from. 
Every time we balance equations, we employ the law of conservation of mass!Ī pure compound will always have the same proportion of the same elements, according to the law of constant composition. 
That means that in a chemical reaction, the amount of each element in the starting materials and the products must be the same. In a closed system, the law of conservation of mass states that matter cannot be created or destroyed. The law of conservation of mass and the law of constant composition were the foundations of Dalton’s hypothesis. 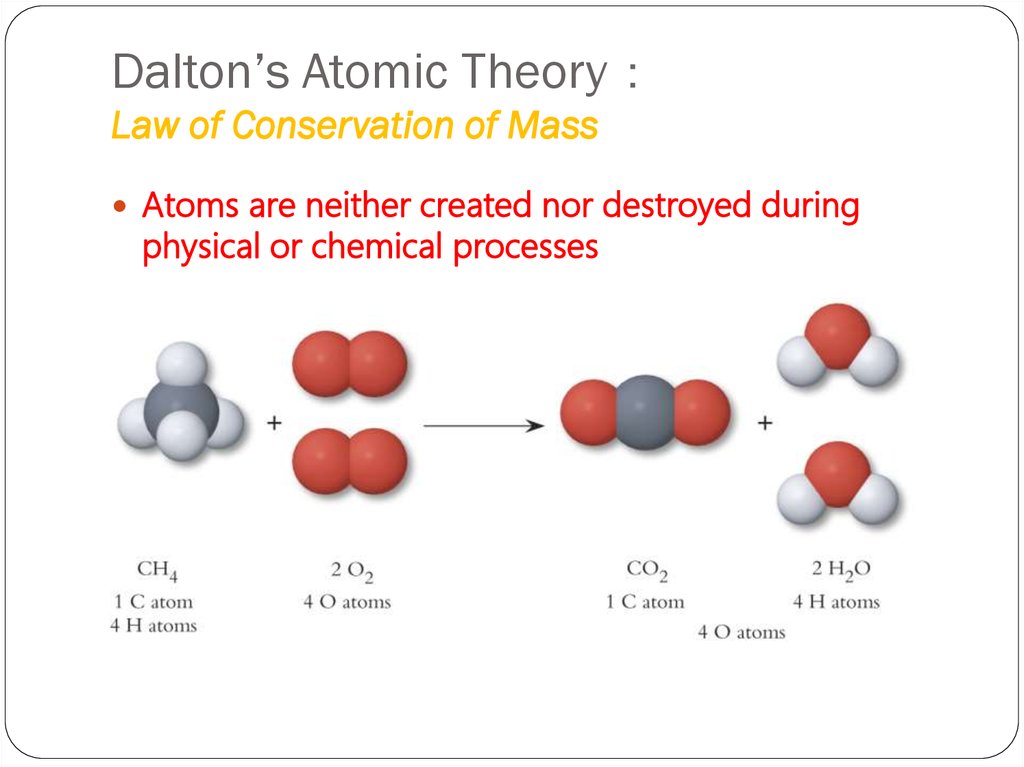
According to the fourth section of the theory, a chemical reaction is an atom rearrangement, and the discovery of subatomic particles and isotopes necessitated certain adjustments to the hypothesis.Compounds are made up of two or more different types of atoms, according to the third part.The theory’s second portion states that all atoms of a given element have the same mass and characteristics.The first component of Dalton’s theory is that all matter is made up of indivisible atoms, as stated by the principles of mass conservation and constant composition.The atomic theory of Dalton was the first effort to characterize all matter in terms of atoms and their properties.All matter, he suggested, is made up of small indivisible particles known as Atoms, which he imagined as “solid, heavy, hard, impenetrable, moving particle(s).” 
The law of conservation of mass and the law of definite proportions, according to Dalton, might be explained using the concept of atoms. He is known for introducing the atomic theory. It is labeled 2 H subscript 2 molecules.John Dalton FRS was an English chemist, physicist and meteorologist. The diagram for 2 H subscript 2 shows two sets of bonded, white spheres. It is labeled one H subscript 2 molecule. The diagram for H subscript 2 shows two white spheres bonded together. The diagram for 2 H shows two white spheres that are not bonded together. The diagram for H shows a single, white sphere and is labeled one H atom. \): The symbols H, 2H, H 2, and 2H 2 represent very different entities.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
